Principle
By flashing the solution in vacuum, the solution temperature is gradually reduced. This reduces the solubility of the salt and causes crystallization. The resulting vapour steam can be used for preheating.
By flashing the solution in vacuum, the solution temperature is gradually reduced. This reduces the solubility of the salt and causes crystallization. The resulting vapour steam can be used for preheating.
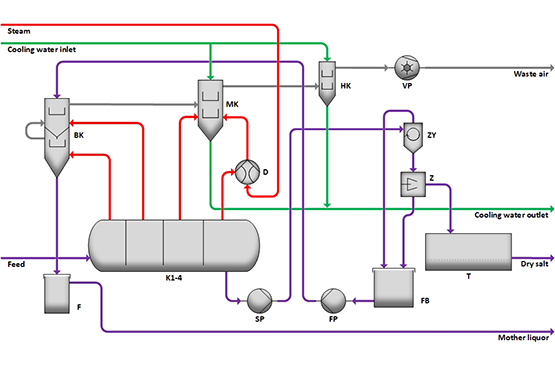
Vacuum cooling crystallization is mainly used for salts with a decreasing solubility at decreasing temperature. A warm, almost saturated solution enters the multi-stage vacuum crystallizer K1-4. In this case, the pressure is gradually reduced, whereby solvent (water) evaporates and the solution cools down to the boiling temperature. By lowering the temperature, salt crystallizes. This is held in suspension by air sucked in at the bottom of the crystallizer (air agitation) and is transported to the outlet. From there, the suspension is pumped to the cyclone ZY for thickening by means of the suspension pump SP. The liquid is then separated in the centrifuge Z. The salt is dried in dryer T. The resulting vapour steam can be used to preheat the mother liquor in the bath condenser BK or condensed with cooling water in the mixing condenser MK. The stage pressure and thus the solution temperature can be reduced even further by use of steam jet pump D. The inert gases are extracted with a vacuum pump VP.